Histamine: Difference between revisions
mNo edit summary |
No edit summary |
||
| Line 124: | Line 124: | ||
==References== | ==References== | ||
<references /> | <references /> | ||
[[Category:Physiology]] | [[Category:Physiology]] | ||
Latest revision as of 20:42, 27 March 2022
Histamine is relevant to musculoskeletal medicine due to its role as a neurotransmitter in the peripheral and central nervous system. It is particularly relevant in neuropathic pain, alloknesis, and the use of antihistamines in neuropathic pain.
Synthesis
Histamine, or 2-(4-imidazolyl)-ethylamine, is synthesised from the amino acid L-histidine through oxidative decarboxylation via catalysis with histidine decarboxylase.
Itch
Itch Clinical Effects
Itch is thought to be a significant aspect of nociception. It has many similarities to pain, but generates a different behavioural response. Pain elicits a withdrawal reflex, while itch elicits a scratch reflex.
Following a stimulus resulting in itch, the individual experience itch at the site which persists briefly after the stimulus is removed. Following this there is a poorly localised area around the stimulus site, which when exposed to gentle touch, elicits intense itch. This is called alloknesis, and is analogous to allodynia.
There are four types of itch
- Pruritoceptive itch, transmitted by C fibres e.g. with inflammation or skin dryness
- Neuropathic itch, the archetypal disease being post-herpetic neuropathic pain. There is a loss of peripheral sensory neurons, which may lead to hyperactivity in the central itch-specific neurons or possibly disinhibit second order neurons.
- Neurogenic itch, occurs without any neural pathology. For example cholestatic itch (through opioid peptides synthesised by the liver), and exogenous opioid agonism on mu receptors.
- Psychogenic itch, such as in delusional parasitosis.
Itch Neurophysiology
Itch is transmitted by a dedicated subclass of C fibres, distinct from the polymodal C fibres involved in pain transmission. These so-called "itch neurons" have extensive terminal branching, slow conduction velocities, and a long lasting response to histamine. Itch neurons are not sensitive to mechanical stimuli.
There are no specific itch receptors on peripheral nerve endings in the skin, unlike what we see with pain. It is thought that there may be such receptors in second order neurons.
In the cerebral cortex, with itch PET imaging has shown co-activation of the anterior cingulate cortex, supplementary motor area, and inferior parietal lobe. The activation pattern is similar to that of pain, but without activation of the thalamic and somatosensory cortex.
The reason why rubbing inhibits the sensation of itch is because this stimulates myelinated A fibre low threshold mechanoreceptors. Scratching also inhibits itch though the stimulation of nociceptors.
Clinical Relevance
Pruritis is seen in many medical conditions such as CKD, lymphoma, HIV, post-herpetic neuralgia, multiple sclerosis, cholestasis, and certain dermatological conditions.
Itch is a common side effect of morphine. This is not an allergic reaction, but rather due to activation of mu receptors. Kappa and delta agonism does not evoke a similar response. Consequently, naltrexone an opioid receptor antagonist, reduces the itch from morphine, but histamine antagonists have no effect.
Histamine Effects
Histamine is pleiotropic, if not the most pleiotropic molecule in the body. It is present in many cells. It is released by both neuronal and non-neuronal cells. It is involved in smooth muscle contraction in the lungs, uterus, and intestine; gastric acid secretion; vasodilation; modulation of heart rate and contractility.
Role in the Immune System
Histamine is a hydrophilic vasoactive amine. It is a potent vasodilator being involved in the acute inflammatory response and plays a key role in many allergic reactions.
Mast cells are recruited to sites of tissue injury, and are also resident in most tissues. They release proinflammatory mediators, including NGF upon activation.
The "triple response of Lewis" is a cutaneous response that occurs from the firm stroking of the skin. The same response occurs with the intradermal injection of histamine.
- Red spot, appears within a few seconds caused by capillary vasodilation.
- Flare, develops slowly as a redness in the surrounding area due to arteriolar dilation mediated by axon reflex.
- Wheal, develops within 1-2 minutes caused by exudation of extracellular fluid from capillaries and venules.
Role in the Nervous System
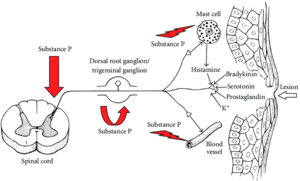
Histamine is a neurotransmitter in both the CNS and PNS. It is involved in multiple homeostatic physiological and pathological processes, for example the sleep-wake cycle, temperature control, memory, pain sensation, synaptic plasticity, and learning.
In experimental models of neuropathic pain, histamine can act as an antinociceptive mediator (analgesic) in the central nervous system, but a nociceptive mediator (algesic) in the peripheral nervous system.[1]
Peripheral Nervous System
There are distinct itch-generating neurons in the peripheral nervous system. In the superficial layers of the skin, the release of histamine from mast cells is a pruritogenic event through the binding of H1 receptors in the terminal membranes of a special subgroup of C-afferent fibres.
There is a bidirectional communication of nerves and mast cells in chemical nociception. Histamine along with multiple other chemicals are released in response to tissue injury. These chemicals stimulate the peripheral endings of polymodal sensory neurons through the TRPV1 receptor. Neurogenic inflammation occurs, which describes the release of inflammatory mediators, especially substance P and calcitonin gene-related peptite, from the peripheral endings of afferent sensory neurons. The inflammatory response that is generated causes local vasodilation, plasma extravasation, leukocyte and platelet adhesion, and mast cell degranulation. Substance P also acts to induce histamine release from adjacent mast cells, which activates further sensory nerve endings. Furthermore, the action potential that is generated travels to the dorsal horn, spreads to other branches, and substance P is also released there.
Histamine also causes the release of prostaglandin E2 which sensitises pain receptors (see NSAIDs). It plays a role in acute inflammation related to mast cell degranulation, but it has an uncertain role in chronic inflammation. At low levels it may cause an itching sensation, and pain at higher levels.
Histamine in low concentrations acting on sensory neurons causes pruritis, while at high concentrations causes pain. Histamine-induced itch can convert to pain in the setting of neuropathic hyperalgesia. In the epidermis histamine causes itching, and in the dermis it causes pain with or without itching.
It is also involved in neuropathic pain in the peripheral nervous system. Following nerve injury, there is an increase in H1 receptor expression in injured afferent nerves, and histamine is released by mast cells. Histamine is a chemoattractant of mast cells, and there is also the recruitment of macrophages and neutrophils.[2]
Post-herpetic neuralgia has some distinct effects. Following partial nerve injury with zoster infection, there may be de novo expression of histamine receptors of primary nociceptive neurons (i.e. not just the itch subclass), as well as central sensitisation to histamine, and synaptic connections between histamine-sensitive C fibres to pain-signalling second order neurons.
Central Nervous System
Histamine injected into various brain areas has either algesic or analgesic effects depending on the dose.
In second order neurons histamine can act and potentiate NMDA receptors.
Histamine Receptors
Histamine exerts its effects primarily by binding to G protein-coupled histamine receptors, designated H1 through H4. Histamine also activates ligand-gated chloride channels in the brain and intestinal epithelium.[3] They vary in the pharmacology and signal transduction properties. Therefore the effect on pain physiology varies depending on the receptor subtype that is activated.
Excitatory histamine receptor activity is associated with increased pain, while inhibition of this activity is associated with neuroprotective and antinociceptive effects.
H1 and H2 receptors are predominantly found post-synaptically. Less is known about H3 and H4 receptors. H3 receptors are found mainly in neurons and are present pre-synaptically, but it is uncertain whether they are present post-synaptically. H4 receptors also require further study; they are not found in large numbers in the nervous system, but may be involved in pain sensitivity modulation. Presynaptic histamine receptors act as autoreceptors or heteroreceptors and provide positive or negative feedback relating to neurotransmitter release from axon terminals.
The table below is from wikipedia.
| G-protein coupled receptor | Location | Function |
|---|---|---|
| H1 receptor | • CNS: Expressed on the dendrites of the output neurons of the histaminergic tuberomammillary nucleus, which projects to the dorsal raphe, locus coeruleus, and additional structures.
• Periphery: Smooth muscle, endothelium, sensory nerves |
• CNS: Sleep-wake cycle (promotes wakefulness), body temperature, nociception, endocrine homeostasis, regulates appetite, involved in cognition
• Periphery: Causes bronchoconstriction, bronchial smooth muscle contraction, urinary bladder contractions, vasodilation, promotes hypernociception (visceral hypersensitivity), involved in itch perception and urticaria. |
| H2 receptor | • CNS: Dorsal striatum (caudate nucleus and putamen), cerebral cortex (external layers), hippocampal formation, dentate nucleus of the cerebellum
• Periphery: Located on parietal cells, vascular smooth muscle cells, neutrophils, mast cells, as well as on cells in the heart and uterus |
• CNS: Not established (note: most known H2 receptor ligands are unable to cross the blood–brain barrier in sufficient concentrations to allow for neuropsychological and behavioral testing)
• Periphery: Primarily involved in vasodilation and stimulation of gastric acid secretion. Urinary bladder relaxation. Modulates gastrointestinal function. |
| H3 receptor | Located in the central nervous system and to a lesser extent peripheral nervous system tissue | Autoreceptor and heteroreceptor functions: decreased neurotransmitter release of histamine, acetylcholine, norepinephrine, serotonin. Modulates nociception, gastric acid secretion, and food intake. |
| H4 receptor | Located primarily on basophils and in the bone marrow. It is also expressed in the thymus, small intestine, spleen, and colon. | Plays a role in mast cell chemotaxis, itch perception, cytokine production and secretion, and visceral hypersensitivity. Other putative functions (e.g., inflammation, allergy, cognition, etc.) have not been fully characterized. |
| Ligand-gated ion channel | Location | Function |
| Histamine-gated chloride channel | Putatively: CNS (hypothalamus, thalamus) and intestinal epithelium | Brain: Produces fast inhibitory postsynaptic potentials
Intestinal epithelium: chloride secretion (associated with secretory diarrhea) |
Resources
- Open access article on the role of histamine in neuropathic pain by Obara et al.[4]
- Open access article on neurogenic inflammation by Rosa et al.[5]
References
- ↑ Yu J, Lou GD, Yue JX, Tang YY, Hou WW, Shou WT, Ohtsu H, Zhang SH, Chen Z. Effects of histamine on spontaneous neuropathic pain induced by peripheral axotomy. Neurosci Bull. 2013 Jun;29(3):261-9. doi: 10.1007/s12264-013-1316-0. Epub 2013 Mar 13. PMID: 23494529; PMCID: PMC5561842.
- ↑ Obara I, Telezhkin V, Alrashdi I, Chazot PL. Histamine, histamine receptors, and neuropathic pain relief. Br J Pharmacol. 2020 Feb;177(3):580-599. doi: 10.1111/bph.14696. Epub 2019 Jun 7. PMID: 31046146; PMCID: PMC7012972.
- ↑ Panula P, Chazot PL, Cowart M, Gutzmer R, Leurs R, Liu WL, Stark H, Thurmond RL, Haas HL. International Union of Basic and Clinical Pharmacology. XCVIII. Histamine Receptors. Pharmacol Rev. 2015 Jul;67(3):601-55. doi: 10.1124/pr.114.010249. PMID: 26084539; PMCID: PMC4485016.
- ↑ Obara et al.. Histamine, histamine receptors, and neuropathic pain relief. British journal of pharmacology 2020. 177:580-599. PMID: 31046146. DOI. Full Text.
- ↑ Rosa, A C, and R Fantozzi. “The role of histamine in neurogenic inflammation.” British journal of pharmacology vol. 170,1 (2013): 38-45. doi:10.1111/bph.12266

