Hypermobile Ehlers Danlos Syndrome
Diagnostic Criteria
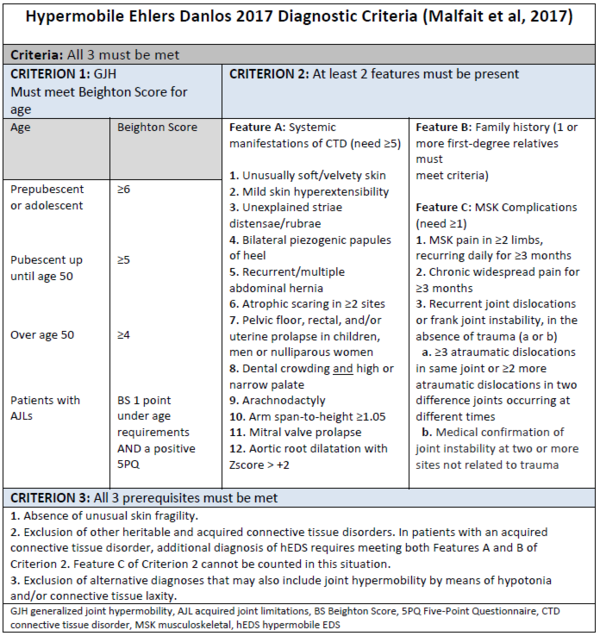
hEDS lies within a spectrum from from asymptomatic joint hypermobility, to “non syndromic” hypermobility spectrum disorder, to hEDS. The latest diagnostic criteria for hEDS were published in 2017 [1]
hEDS Diagnostic Criteria Checklist
| Motion | Negative | Unilateral | Bilateral |
|---|---|---|---|
| Passive dorsiflexion of the 5th finger >90 degrees | 0 | 1 | 2 |
| Passive flexion of the thumbs to the forearms | 0 | 1 | 2 |
| Hyperextension of the elbows >10 degrees | 0 | 1 | 2 |
| Hyperextension of the knees >10 degrees | 0 | 1 | 2 |
| Forward flexion of the trunk with knees fully extended and palms resting on the floor | 0 | 1 | |
Must meet Beighton Score for age
| Age | Beighton Score |
|---|---|
| Prepubescent or adolescent | ≥6 |
| Pubescent up until age 50 | ≥5 |
| Over age 50 | ≥4 |
| Patients with Acquired joint limitations | BS 1 point under age requirements AND a positive 5PQ |
| Five Point Questionnaire for identifying hypermobility (5PQ) |
|---|
| Criteria: Two or more “yes” answers suggest joint hypermobility. |
|
Feature A: Systemic manifestations of CTD (need ≥5)'
- Unusually soft/velvety skin
- Mild skin hyperextensibility
- Unexplained striaedistensae/rubrae
- Bilateral piezogenic papules of heel
- Recurrent/multiple abdominal hernia
- Atrophic scaring in ≥2 sites
- Pelvic floor, rectal, and/or uterine prolapse in children, men or nulliparous women
- Dental crowding and high or narrow palate
- Arachnodactyly
- Arm span-to-height ≥1.05
- Mitral valve prolapse
- Aortic root dilatation with Z score > +2
Skin extensibility as measured by pinching and lifting the cutaneous and subcutaneous layers of the skin on the volar surface at the middle of the non-dominant forearm as described in Remvig et al, 2009. Skin extensibility of >1.5 cm is considered the upper end of normal. It is likely that the hyperextensibility of the skin in hEDS overlaps significantly with that of “normal” skin. Therefore, extensibility of more than 1.5 cm is “positive.” If extensibility >2.0 cm is present especially in combination with other cutaneous features, such as papyraceous scars, molluscoid pseudotumors and/or subcutaneous spheroids, consider other EDS types as possible alternative diagnoses (mainly cEDS and classical-like EDS)
Feature B: Family history (1 or more first-degree relatives must meet criteria)
Feature C: MSK Complications(need ≥1)
- MSK pain in ≥2 limbs, recurring daily for ≥3 months
- Chronic widespread pain for ≥3 months
- Recurrent joint dislocations or frank joint instability, in the absence of trauma (a or b)
- ≥3 atraumatic dislocations in same joint or ≥2 more atraumatic dislocations in two difference joints occurring at different times
- Medical confirmation of joint instability at two or more sites not related to trauma
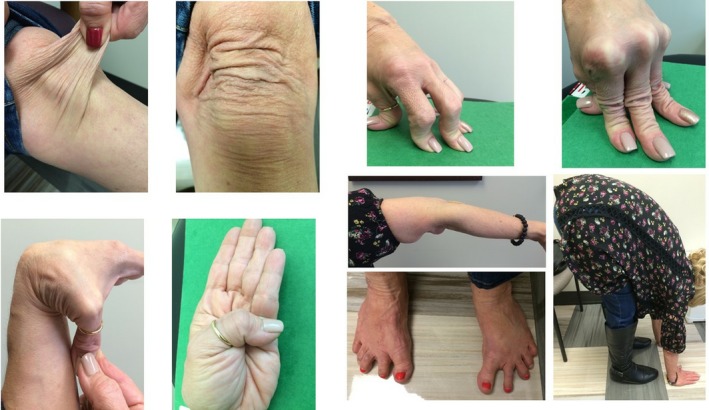
Skin hyperextensibility and Generalized Joint Hypermobility on physical exam[2]
- Absence of unusual skin fragility.
- Exclusion of other heritable and acquired connective tissue disorders. In patients with an acquired onnective tissue disorder, additional diagnosis of hEDS requires meeting both Features A and B of Criterion 2. Feature C of Criterion 2 cannot be counted in this situation.
- Exclusion of alternative diagnoses that may also include joint hypermobility by means of hypotonia and/or connective tissue laxity
Controversies
Controversy of hEDS vs Fibromyalgia vs BJHS
The first problem is that the Beighton scoring method for assessing generalised hypermobility is highly flawed and has garnered some controversy. Some authors argue for a greater emphasis on clinician judgement.[3]
The hEDS subtype of EDS has varying levels of acceptance and awareness as a diagnostic entity by various relevant specialists in New Zealand. What some clinicians diagnose as hEDS, some clinicians may instead diagnose Fibromyalgia, another controversial condition that is not commonly diagnosed in Musculoskeletal Medicine, benign joint hypermobility syndrome plus fibromyalgia, or simply Central Sensitisation which again has controversies.
The evidence for applying any one or more of the above diagnoses is not robust. Some of the controversy stems from the lack of clearly objective tests. hEDS, fibromyalgia, Benign Joint Hypermobility Syndrome, and central sensitisation are all diagnosed using subjective "committee criteria" or "questionnaires." Committee diagnoses are when a group of experts come together to publish a list of symptoms and signs, and that if you fulfill those symptoms you have the disorder. There is always a but; committee diagnoses always stipulate that other conditions must be excluded first. Therefore it is not logically possible to choose between one committee diagnosis and the other (e.g. hEDS and fibromyalgia) when the patient fulfills the criteria for both conditions for the same symptoms. In the absence of widely accepted objective test and the removal of the "exclude other conditions" stipulation, who is to say that hEDS trumps fibromyalgia, or that fibryomyalgia trumps hEDS? Furthermore how does one accurately differentiate between primary chronic widespread pain versus pain with joint laxity?
The existence of a committee diagnosis for a condition does not in itself mean that the condition doesn't exist. Migraine, or the entire field of psychiatric diagnoses are good examples of mainstream committee diagnoses that have broad acceptance. Glioblastoma and myocardial infarction are good examples of a more objective diagnoses. The subjective nature of diagnosis can lead to poor inter-observer reliability and can fuel the flame of controversy.
There is some conflict between Benign Joint Hypermobility Syndrome (BJHS) and hypermobile Ehlers Danlos Syndrome. The former is diagnosed using the 1998 Brighton criteria and has come from the Rheumatology literature, which is probably why it is favoured by some Rheumatologists. Hypermobile Ehlers Danlos Syndrome is diagnosed using the 2017 diagnostic criteria and comes from the Genetics and Paediatrics literature. Some authors and clinicians believe that these two conditions are in fact the same thing.
Patients in whom this or related diagnoses are being considered are clearly suffering and have very real symptoms. Clinicians may of course make such diagnoses, and indeed they do so regularly. Making a committee diagnosis can be helpful in framing and providing a narrative through which to support management in a patient centered model of care. Medical discoveries are all made through trying new things, and as long as the patient is fully informed then the clinician and patient may wish to explore hEDS as a framework.
However at the extreme it can lead to polypharmacy, perpetuate the cycle of hope and despair, and the patient may experience resentment from some health care providers who do not "believe" in the disorder. One should always keep in mind the limitations of the diagnostic criteria and treatment evidence, the socio-psychological factors, the potential benefits and harms of making the diagnosis, and the context of the broader healthcare system.
Controversy of "Rare" Label
hEDS as it is currently diagnosed is not rare. The prevalence in a UK study was 1 in 500,[4] which is four times more common than the generally accepted 1 in 2000 for a rare disease. After decades of looking there has been no single genetic defect found in the majority of cases. It is highly improbable that a single gene will be found at this point. hEDS behaves exactly like a common complex disease that has both polygenetic and environmental influences like asthma, type 2 diabetes, and fibromyalgia. All of these common conditions also run in families. For example Type 2 diabetes has an heritability factor of 30-70%.[5]
Psychosomatic Label
There is no evidence that hEDS is psychosomatic or a conversion disorder. Essentially, hEDS is something akin to a state where hypermobility is a driver for central changes in sensory processing in a person that is genetically and environmentally predisposed to such central changes. The main difference to fibromyalgia is that nociceptive pain is more common and there may be a role for treating any peripheral pain drivers as well as central factors. The psychological distress which is without a doubt very common as a comorbidity does not cause the pain but may amplify it. More importantly is that the disability drives the psychological distress, and if the disability can be improved then usually the distress also improves.
Triad of hEDS, MCAS, and POTS
Some patients with hEDS may also be diagnosed with mast cell activation syndrome (MCAS) and postural orthostatic tachycardia syndrome (POTS). Kohn et al evaluated the literature behind each of these three conditions, and the relationship between them[6]. They concluded that there is no proven relationship between hypermobile Ehlers Danlos Syndrome, Mast Cell Activation Syndrome, and POTS, and no proven pathophysiological mechanism linking them. The main limitation of their review was that most of the studies to date used the older criteria of hEDS which was much more inclusive and more vague, making analysis more difficult. They theorised that the reason that these conditions have been associated is because there is a large overlap of non-specific symptomatology, summarised in their Venn Diagram below. Please note that MCAS as a diagnostic entity is controversial and only minority support in New Zealand.[7]
Differential Diagnosis
- Generalised Joint Hypermobility
- Hypermobility Spectrum Disorder or Hypermobile Ehlers Danlos Syndrome
- Ehlers Danlos Syndrome
- Marfan Syndrome
- Osteogenesis Imperfecta Type 1
- Geroderma Osteodysplasticum
- Loeys-Dietz
- Arterial Tortuosity Syndrome
- Lateral Meningocele Syndrome
- Bethlem Myopathy
- Ullrich congenital muscular dystrophy
- Acromesomelic Dysplasia
- Coffin-Lowry Syndrome
- Cohen Syndrome
- Cranioectodermal Dysplasia
- Deletion 2q37 Syndrome
- Down Syndrome
- Floating-Harbor Syndrome
- Hajdu-Cheney Syndrome
- Kabuki Syndrome
- Loeys-Dietz Syndrome
- Macrocephaly-Capillary Malformation
- Meier-Gorlin Syndrome
- Metatropic Dysplasia
- Microcephalic Primordial Dwarfing Syndrome
- Microdeletion 15q24 Syndrome
- Microdeletion 22q11.2 Syndrome
- Morquio Syndrome
- Multiple Endocrine Neoplasia Type 2B
- Peters'-Plus Syndorme
- Pitt-Hopkins Syndrome
- Pseudoachondroplasia
- SHORT Syndrome
- Stickler Syndrome
- 3-M Syndrome
- XXXY and XXXXY SDyndrome
Resources
- Dr Matthew Preston | Ehlers-Danlos Syndromes (matpre.nz)
- Ehlers-Danlos Syndrome (EDS) Algorithm and Resources for Primary Care - Mountain States GeneticsMountain States Genetics
- The Ehlers-Danlos syndromes (EDS) GP Toolkit
- Special Issue in American Journal of Medical Genetics December 2021
References
Papers of particular interest have been highlighted as: ◆ of special interest ◆◆ of outstanding interest
- ↑ ◆◆ Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet. 2017;175(1):8-26. doi:10.1002/ajmg.c.31552
- ↑ Zhang et al.. Hypermobile type Ehlers-Danlos syndrome associated with hypogammaglobulinemia and fibromyalgia: A case-based review on new classification, diagnosis, and multidisciplinary management. Clinical case reports 2019. 7:680-685. PMID: 30997063. DOI. Full Text.
- ↑ ◆ Malek, S., Reinhold, E.J. & Pearce, G.S. The Beighton Score as a measure of generalised joint hypermobility. Rheumatol Int 41, 1707–1716 (2021). https://doi.org/10.1007/s00296-021-04832-4
- ↑ Demmler, Joanne C.; Atkinson, Mark D.; Reinhold, Emma J.; Choy, Ernest; Lyons, Ronan A.; Brophy, Sinead T. (2019-11-04). "Diagnosed prevalence of Ehlers-Danlos syndrome and hypermobility spectrum disorder in Wales, UK: a national electronic cohort study and case-control comparison". BMJ open. 9 (11): e031365. doi:10.1136/bmjopen-2019-031365. ISSN 2044-6055. PMC 6858200. PMID 31685485.
- ↑ Laakso, Markku; Fernandes Silva, Lilian (2022-08-04). "Genetics of Type 2 Diabetes: Past, Present, and Future". Nutrients. 14 (15): 3201. doi:10.3390/nu14153201. ISSN 2072-6643. PMC 9370092. PMID 35956377.
- ↑ Kohn A, Chang C. The Relationship Between Hypermobile Ehlers-Danlos Syndrome (hEDS), Postural Orthostatic Tachycardia Syndrome (POTS), and Mast Cell Activation Syndrome (MCAS). Clin Rev Allergy Immunol. 2020;58(3):273-297. doi:10.1007/s12016-019-08755-8
- ↑ Valent, Peter et al. “Mast cell activation syndrome: Importance of consensus criteria and call for research.” The Journal of allergy and clinical immunology vol. 142,3 (2018): 1008-1010. doi:10.1016/j.jaci.2018.06.004
Literature Review
- Reviews from the last 7 years: review articles, free review articles, systematic reviews, meta-analyses, NCBI Bookshelf
- Articles from all years: PubMed search, Google Scholar search.
- TRIP Database: clinical publications about evidence-based medicine.
- Other Wikis: Radiopaedia, Wikipedia Search, Wikipedia I Feel Lucky, Orthobullets,