Opioids
Opioids are an analgesic, but have fell out of favour in the treatment of chronic pain. They currently only have a role in acute pain and chronic cancer related pain.
Classification
Classification by chemistry
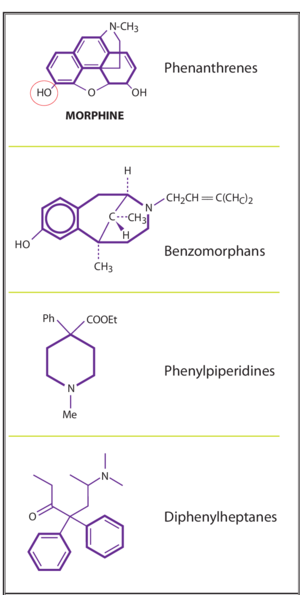
There are four base molecules that give rise to four families of opioids
- Phenathrenes - prototypical opioids such as morphine, codeine, oxycodone
- Diphenylheptanes- methadone and propoxyphene
- Phenylpiperidines - pethidine, fentanyl, and other fentayl related compounds
- Benzomorphans - pentazocine only
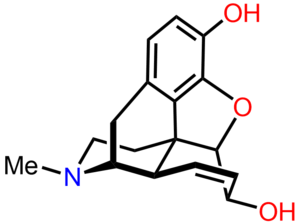
Morphine is the archetypal opioid. It has a benzene ring with a phenolic hydroxyl group at position 3, as well as an alcohol hydroxyl groups at position 6 and at the nitrogen atom. Both of the position 3 and 6 hydroxyl groups can be converted to ethers or esters forming other compounds. The nitrogen group is in a tertiary form and this is important to its analgesic properties.
Tramadol doesn't fit into the above classes. It is an atypical opioid with partial mu agonist activity and central CABA, catecholamine, and serotonergic activities.
Classification by receptor affinity
- Opioid Agonists - Morphine, Codeine, Meperidine, Fentanyl, Sufentanil, Remifentanil, Methadone, Tramadol
- Opioid Agonist/Antagonists & Partial Agonists - Pentazocine, Nalbuphine, Butorphanol, Buprenorphine
- Opioid Antagonists - Nalorphine, Naloxone, Naltrexone, Naltrindole, Nalmefene
Classification by Exogenous Opioids Strength
According to the strength or potency based on the plasma concentrations at which they exert their effects. C50 or the plasma concentration causing a 50% effect.
- Strong Opioids include fentanyl, sufentanil, and remifentanil
- Weak opioids include codeine and tramadol
- Intermediate group includes morphine, methadone, oxycodone, and buprenorphine
Classification by synthetic process
- Naturally occurring compounds - Morphine, Codeine, Thebaine, Papaverine
- Semi-synthetic compounds - Diamorphine (heroin), Dihydromorphone, Buprenorphine, Oxycodone
- Synthetic compounds - Pethidine, Fentanyl, Methadone, Alfentanil, Remifentanil, Tapentadol
Mechanism of Action
Opioid receptors are found within the CNS as well as in peripheral tissues. Most opioids with clinical relevance have primary agonist activity on mu receptors, and so are termed mu agonists. With increasing dose, analgesia theoretically occurs in a log linear fashion. While opioid agonists/antagonists and partial agonists have a ceiling effect.
| Mu (µ) | Delta (δ) | Kappa (κ) | |
| Mu1 - Supraspinal analgesia
Mu2 - Sedation, vomiting, respiratory depression, pruritis, euphoria, anorexia, urinary retention, physical dependence. |
Analgesia, spinal analgesia. Possible psychomimetic and dysphoric effects | Spinal analgesia, sedation, dyspnoea, psychomimetic effects, miosis, respiratory depression, euphoria, dysphoria, dyspnoea | |
| Endogenous Peptides | |||
|---|---|---|---|
| Enkephalins | Agonist | Agonist | |
| β-Endorphin | Agonist | Agonist | |
| Dynorphin A | Agonist | Agonist | |
| Agonists | |||
| Morphine | Agonist | Weak Agonist | |
| Codeine | Weak Agonist | Weak Agonist | |
| Fentanyl | Agonist | ||
| Meperidine | Agonist | Agonist | |
| Methadone | Agonist | ||
| Antagonists | |||
| Naloxone | Antagonist | Weak Antagonist | Antagonist |
| Naltrexone | Antagonist | Weak Antagonist | Antagonist |
Opioids can also antagonise NDMA receptors, thereby activating the descending serotonin and noradrenaline pain pathways. NMDA receptors are thought to be involved in neuropathic pain and the development of tolerance.
While opioids act on opioid receptors but aren't naturally occurring substances in the body. Endogenous opioids or endorphins are the natural ligands and are found in the central nervous system. The first endogenous opioid discovered was enkephalin, but there are also endorphins, dynorphins, and nociceptins. They function as neurotransmitters, and may have a role with hormone secretion, thermoregulation, and cardiovascular control.
Mechanisms of Analgesia
Specific Opioids
Morphine
- Low lipid solubility
- Slow onset action (slow BBB penetration)
- High 1st pass metabolism (LIVER)
- Metabolism CYP450 / glucuronidation
- 40-60% reach systemic circulation
- Renal excretion
Methadone
- Longer duration action
- Limited first pass metabolism
- High bioavailability
- ANTAGONIST NMDA receptor – Neuropathic pain
- Inhibits re-uptake NA/Serotonin
- Metabolised in Liver / excretes feaces (mostly)
Tramadol
- Phenylpiperidine analogue (Codeine)
- Modulation Serotonin and NA reuptake
- Less Respiratory depression
- Less GIT S/E (for comparable analgesic effect)
Tapentadol
- MOP receptor agonist
- NA reuptake inhibition
- Comparable opioid potency to Oxycodone
Ramifentanyl
- High Lipid solubility
- Rapid extra-hepatic metabolism
Codeine
- “Pro-drug”
- Converted to morphine for effect
- 5-10% population unable to convert / ineffective
Pethidine (Meperidine)
- Relative weak opioid (10% morphine)
- ++ anticholinergic effects
- Neurotoxic S/E (Particularly in renal disease)
- Delerium
- Excitation
- Seizures
Opioid Potency Comparison
| Opioid Agonists | Potency | Half-life |
|---|---|---|
| Morphine | 1 | 2 - 3.5 |
| Hydromorphone | 5 - 8 | 2 - 3 |
| Oxycodone | 1.5 | 2 - 3 |
| Methadone | 1 | 24 |
| Fentanyl | 100 | 3 - 4 |
| Codeine | 0.13 | 3 |
| Tramadol | 0.2 | |
| Sufentanil | 1000 | |
| Alfentanil | 20 | |
| Remifentanil | 100 |
| Converting | Ratio | Example | ||
|---|---|---|---|---|
| from | to | |||
| Codeine oral | morphine oral | 10:1 | Codeine 60mg oral | -> morphine 6mg oral |
| Tramadol oral | morphine oral | 10:1 | Tramadol 100mg oral | -> morphine 10mg oral |
| Dihydrocodeine oral | Morphine oral | 10:1 | Dihydrocodeine 100mg oral | -> Morphine 10mg oral |
| Oxycodone oral | Morphine oral | 1:1.5 | Oxycodone 10mg oral | -> Morphine 15mg oral |
| Morphine oral | Oxycodone oral | 2:1 | Morphine 20mg oral | -> Oxycodone 10mg oral |
| Morphine oral | Morphine subcut | 2 to 3:1 | Morphine 30mg oral | -> Morphine 10-15mg subcut |
| Oxycodone oral | Oxycodone subcut | 2:1 | Oxycodone 20mg oral | -> Oxycodone 10mg subcut |
| Oxycodone subcut | Morphine subcut | 1:1 | Oxycodone 10mg subcut | -> Morphine 10mg subcut |
| Morphine oral | Fentanyl subcut | 100:1 | Morphine 30mg oral | -> Fentanyl 0.3mg subcut |
Calculation of oral Morphine Equivalent Daily Dose (oMEDD)
oMEDD (mg) = Current Opioid Dose x Conversion factor
Renal Failure
| % Normal Dose | Dose (mg) | Dose Interval (hourly) | |
|---|---|---|---|
| Mild renal impairment (GFR 20-50) | 75% | 2.5-5 | 6 |
| Moderate renal impairment (10-20) | 50% | 2.5-5 | 6-8 |
| Severe renal impairment (<10) | Use small doses | 1.25-2.5 | 8-12 |
| % Normal Dose | Dose (mg) | Dose Interval (hourly) | |
|---|---|---|---|
| Mild renal impairment (GFR 20-50) | 100% | 50-100 | 6 |
| Moderate renal impairment (10-20) | 50% | 50-100 | 6-8 (modify as needed) |
| Severe renal impairment (<10) | 50% | 50 | 6-8 (modify as needed) |
| % Normal Dose | Dose (mg) | Dose Interval (hourly) | |
|---|---|---|---|
| Mild renal impairment (GFR 20-50) | 50% | 2.5-5 | 6 |
| Moderate renal impairment (10-20) | 25-50% | 2.5-5 | 6-8 (modify as needed) |
| Mild renal impairment (<10) | 25-50% | 2.5-5 | 8-12 (modify as needed |
Fentanyl in Renal Failure
Fentanyl is metabolised in the liver to inactive metabolites. It is a useful Strong opioid in renal failure with stable pain. Fentanyl is available in patches.
Liver Failure
There is variable time of onset and analgesic efficacy. They are reasonably well tolerated in adjusted dosing. Ramifentanyl has no hepatic metabolism. Fentanyl patches are a good choice.
Variation in Sensitivity
Polymorphism in human OPRM1 gene which encodes the mu opoid receptor might contribute to variation in sensitivity.
Adverse Effects
- See also: Opioid Deprescribing
The evidence does not support the use of long term opioid use in chronic non-malignant pain. There are neurohumoral effects which include hormonal dysregulation, immunosuppression, increased risk of falls, impairment of sleep quality, and opioid induced hyperalgesia.