Sodium Channelopathies
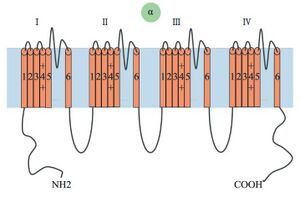
Voltage gated sodium channels are transmembrane polypeptides. The α-subunits are made up of four domains (I-IV). Each domain has six helical transmembrane segments that span the lipid bilayer (S1-6). S1-4 control the gating, S4 senses voltage, while S5 and S6 form hydrophobic pore for ion conduction. There is also an ancillary beta unit for anchoring and kinetics.
Sodium channels can be in one of three states, closed, open, and inactivated.
They are vital for the generation of action potentials (See also Basic Neurophysiology and Channelopathies and Chronic Pain).
Classification
Disease associated mutations have been identified for all major voltage-gated ion channels (Na+, K+, Ca2+, Cl-) and several ligand gated channels (incl. Ach, Gly, GABA). The sodium channelopathies are amongst the most well researched.
There are nine known members of sodium channels labelled Nav1.1 through Nav1.9. The associated genes are named SCN1A through SCN11A (the SCN6/7A gene is part of the Nax sub-family and has uncertain function)
| Protein name | Gene | Expression profile | Function | Associated channelopathies |
|---|---|---|---|---|
| Nav1.1 | SCN1A | Central neurons, [peripheral neurons] and cardiac myocytes | febrile epilepsy, GEFS+, Dravet syndrome (also known as severe myclonic epilepsy of infancy or SMEI), borderline SMEI (SMEB), West syndrome (also known as infantile spasms), Doose syndrome (also known as myoclonic astatic epilepsy), intractable childhood epilepsy with generalized tonic-clonic seizures (ICEGTC), Panayiotopoulos syndrome, familial hemiplegic migraine (FHM), familial autism, Rasmussens's encephalitis and Lennox-Gastaut syndrome | |
| Nav1.2 | SCN2A | Central neurons, peripheral neurons | inherited febrile seizures, epilepsy, and autism spectrum disorder | |
| Nav1.3 | SCN3A | Central neurons, DRG, peripheral neurons and cardiac myocytes | Rapid activation and inactivation, 3x faster
recovery after hyperpolarization than Nav1.7[2] |
epilepsy, pain, brain malformations, ?trigeminal neuralgia |
| Nav1.4 | SCN4A | Skeletal muscle | Certain non-dystrophic myotonias: Sodium channel myotonia, Paramyotonia congenita, Hyperkalaemic periodic paralysis | |
| Nav1.5 | SCN5A | Cardiac myocytes, uninnervated skeletal muscle, central neurons, gastrointestinal smooth muscle cells and Interstitial cells of Cajal | Cardiac: Long QT syndrome Type 3, Brugada syndrome, progressive cardiac conduction disease, familial atrial fibrillation and idiopathic ventricular fibrillation;
Gastrointestinal: Irritable bowel syndrome; | |
| Nav1.6 | SCN8A | Central neurons, dorsal root ganglia, peripheral neurons, heart, glia cells | Epilepsy, ataxia, dystonia, tremor | |
| Nav1.7 | SCN9A | Dorsal root ganglia, sympathetic neurons, peripheral terminals of sensory neurons, axons, central terminals of nociceptors, Schwann cells, and neuroendocrine cells, | Produces a substantial ramp current in response to small slow depolarisation, determining nociceptor terminal threshold. May regulate neurotransmitter release at central terminals. Rapid activation and inactivation. | Gain of function: SCN9A neuropathic pain syndromes: erythromelalgia,Small fibre neuropathy, paroxysmal extreme pain disorder
Loss of function: Congenital Insensitivity to Pain (CIP) a.k.a hereditary sensory and autonomic neuropathy type 2D (HSAN2D) |
| Nav1.8 | SCN10A | Dorsal root ganglia | crucial for transmission of nociceptive information carrying most of the depolarising phase of the action potential in the DRG following initiation by Nav1.7. Rapid activation and slow inactivation. | Small fibre neuropathy, familial episodic pain syndrome type 2, autosomal recessive neuromuscular disease and epileptic encephalopathy, Brugada syndrome, and congenital abnormalities of the kidney and urinary tract. |
| Nav1.9 | SCN11A | Dorsal root ganglia | slow gating kinetics generating persistent currents at near-resting membrane potentials and so are important in regulation of membrane excitability. | Small fibre neuropathy, familial episodic pain syndrome type 3, autosomal dominant hereditary sensory and autonomic neuropathy type 7 (HSAN7) |
| Nax | SCN7A | heart, uterus, skeletal muscle, astrocytes, dorsal root ganglion cells | none known |
Clinical Features
Dravet Syndrome (SCN1A)
Sodium channels produce action potentials, so this raises the question as to why loss of function would result in increased neural activity. The explanation lies within understanding that Nav1.1 is expressed primarily in GABAergic inhibitory neurons. So reduced activity in inhibitory neurons results in an excitatory-inhibitory imbalance (EI imbalance) favouring excitatory impulses.
While this disease is not at face value relevant to Musculoskeletal Medicine, it is a useful learning point because it shows the importance of the EI balance. EI imbalance has been implicated in non genetic conditions such as head injury whereby there may be preferential injury to the inhibitory neurons and/or preferential post-injury sprouting of excitatory neurons.
Nondystrophic Myotonias
The primary sodium channel in skeletal muscle is Nav1.4 (SCN4A). There are two different conditions caused by mutations in this ion channel
- Paramyotonia congenita – the pathogenic variant results in slowing of the fast inactivation process. The channels stay open for too long resulting in repetitive action potentials and prolonged contractions. Episodic generalised stiffness in early childhood and pain. Worsens with repeated contractions, relieved by rest.
- Hyperkalaemic periodic paralysis – mutations that cause impairment of slow inactivation process. The muscles are predisposed to be depolarised, have action potential block, and therefore paralysis
Primary (SCN9A) Erythromelalgia
Primary Erythromelalgia is characterized by recurrent, bilateral episodes of intense burning pain, redness, warmth, and sometimes swelling, SCN9A-EM most commonly affects the feet. However, severely affected individuals may also experience involvement of the hands, legs, arms, face, and/or ears.
Paroxysmal Extreme Pain Disorder
Paroxysmal Extreme Pain Disorder typically begins during infancy or the neonatal period and includes autonomic symptoms such as skin flushing, patchy or asymmetric colour changes (harlequin), non-epileptic tonic attacks (stiffening), and fainting accompanied by a slow heart rate (syncope with bradycardia). Later manifestations involve severe, deep burning pain in the rectal, ocular, or submandibular regions, along with erythematous skin changes (flushing).
Inherited Small Fibre Neuropathy
Small Fibre Neuropathy is an adult-onset condition marked by neuropathic pain in a stocking and glove distribution, often described as burning. Autonomic symptoms may include dry eyes and mouth, orthostatic dizziness, palpitations, and bowel or bladder disturbances. Large nerve fiber functions, such as normal strength, tendon reflexes, and vibration sense, are preserved.
See Also
GeneReviews - SCN9A Pain Conditions
Resources
References
- ↑ "Sodium channel". Wikipedia (in English). 2022-12-02.
- ↑ Cummins, Theodore R.; Sheets, Patrick L.; Waxman, Stephen G. (2007-10). "The roles of sodium channels in nociception: Implications for mechanisms of pain". Pain. 131 (3): 243–257. doi:10.1016/j.pain.2007.07.026. ISSN 1872-6623. PMC 2055547. PMID 17766042. Check date values in:
|date=(help)
Literature Review
- Reviews from the last 7 years: review articles, free review articles, systematic reviews, meta-analyses, NCBI Bookshelf
- Articles from all years: PubMed search, Google Scholar search.
- TRIP Database: clinical publications about evidence-based medicine.
- Other Wikis: Radiopaedia, Wikipedia Search, Wikipedia I Feel Lucky, Orthobullets,